Nanoparticle size and surface chemistry determine serum protein adsorption and macrophage uptake
Carl D Walkey, Jonathan B Olsen, Hongbo Guo, Andrew Emili, Warren CW Chan
J. Am. Chem. Soc. 2012, 134, 4, 2139–2147 | DOI: 10.1021/ja2084338
Abstract
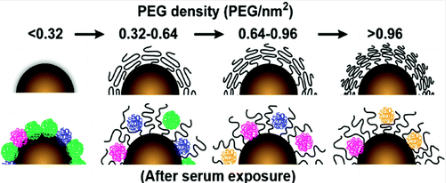
Delivery and toxicity are critical issues facing nanomedicine research. Currently, there is limited understanding and connection between the physicochemical properties of a nanomaterial and its interactions with a physiological system. As a result, it remains unclear how to optimally synthesize and chemically modify nanomaterials for in vivo applications. It has been suggested that the physicochemical properties of a nanomaterial after synthesis, known as its “synthetic identity”, are not what a cell encounters in vivo. Adsorption of blood components and interactions with phagocytes can modify the size, aggregation state, and interfacial composition of a nanomaterial, giving it a distinct “biological identity”. Here, we investigate the role of size and surface chemistry in mediating serum protein adsorption to gold nanoparticles and their subsequent uptake by macrophages. Using label-free liquid chromatography tandem mass spectrometry, we find that over 70 different serum proteins are heterogeneously adsorbed to the surface of gold nanoparticles. The relative density of each of these adsorbed proteins depends on nanoparticle size and poly(ethylene glycol) grafting density. Variations in serum protein adsorption correlate with differences in the mechanism and efficiency of nanoparticle uptake by a macrophage cell line. Macrophages contribute to the poor efficiency of nanomaterial delivery into diseased tissues, redistribution of nanomaterials within the body, and potential toxicity. This study establishes principles for the rational design of clinically useful nanomaterials.