Nanoparticle–blood interactions: the implications on solid tumour targeting
James Lazarovits, Yih Yang Chen, Edward A Sykes, Warren CW Chan
Chem. Commun., 2015, 51, 2756-2767 | DOI: 10.1039/C4CC07644C
Abstract
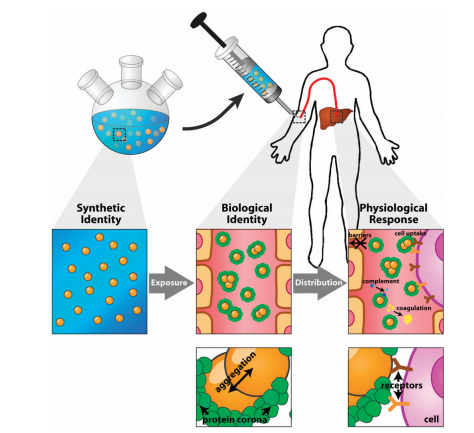
Nanoparticles are suitable platforms for cancer targeting and diagnostic applications. Typically, less than 10% of all systemically administered nanoparticles accumulate in the tumour. Here we explore the interactions of blood components with nanoparticles and describe how these interactions influence solid tumour targeting. In the blood, serum proteins adsorb onto nanoparticles to form a protein corona in a manner dependent on nanoparticle physicochemical properties. These serum proteins can block nanoparticle tumour targeting ligands from binding to tumour cell receptors. Additionally, serum proteins can also encourage nanoparticle uptake by macrophages, which decreases nanoparticle availability in the blood and limits tumour accumulation. The formation of this protein corona will also increase the nanoparticle hydrodynamic size or induce aggregation, which makes nanoparticles too large to enter into the tumour through pores of the leaky vessels, and prevents their deep penetration into tumours for cell targeting. Recent studies have focused on developing new chemical strategies to reduce or eliminate serum protein adsorption, and rescue the targeting potential of nanoparticles to tumour cells. An in-depth and complete understanding of nanoparticle–blood interactions is key to designing nanoparticles with optimal physicochemical properties with high tumour accumulation. The purpose of this review article is to describe how the protein corona alters the targeting of nanoparticles to solid tumours and explains current solutions to solve this problem.