An analysis of the binding function and structural organization of the protein corona
Yuwei Zhang, Jamie LY Wu, James Lazarovits, Warren CW Chan
J. Am. Chem. Soc. 2020, 142, 19, 8827–8836 | DOI:10.1021/jacs.0c01853
Abstract
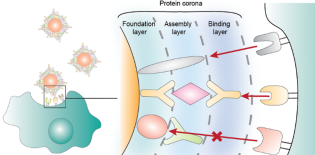
Blood proteins adsorb onto the surface of nanoparticles after intravenous injection to form a protein corona. The underlying organization and binding function of these adsorbed proteins remain unclear. This can impact how the corona mediates cell and tissue interactions. Here, we investigated the function and structural organization of the protein corona using an immunoassay approach. We discovered that only 27% of the adsorbed proteins examined are functional for binding to their target protein. This is because the corona architecture is not a monolayer, but an assembly of proteins that are bound to each other. We further demonstrated that we can control the binding functionality of a protein by changing the organization of proteins in the assembly. We show that manipulation of the corona protein composition and assembly can influence their interactions with macrophage cells in culture. This study provides detailed functional and structural insights into the protein corona on nanomaterials and offers a new strategy to manipulate it for controlled interactions with the biological system.